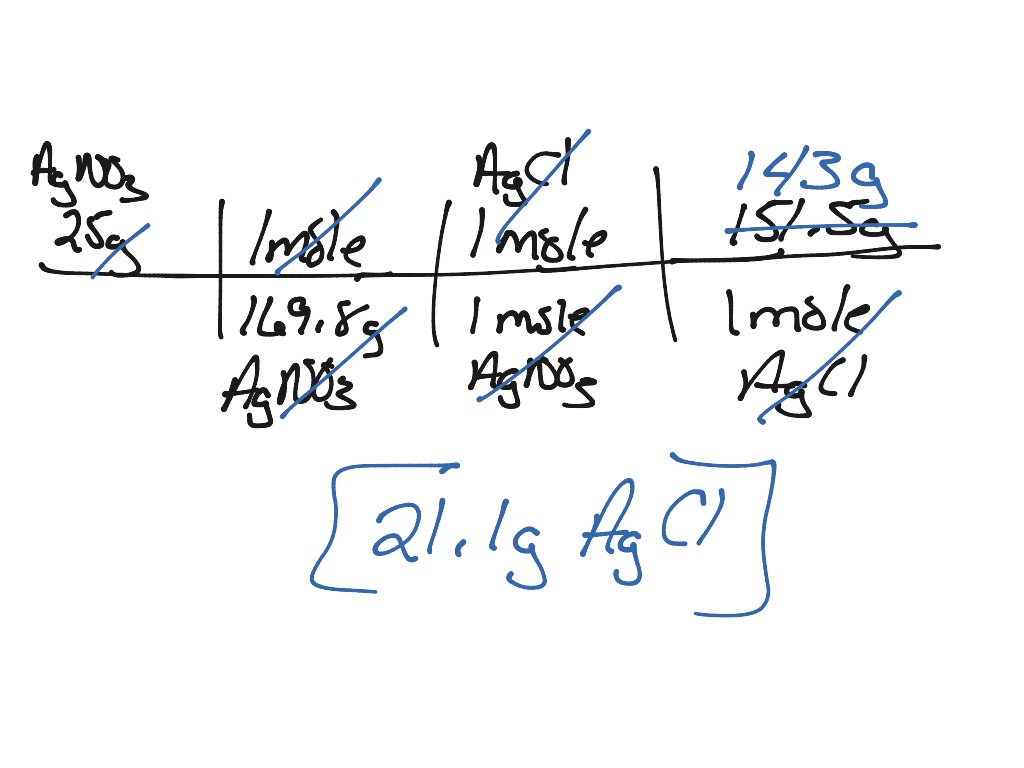 
For example, a saturated solution of silver chloride is one in which the equilibrium shown below has been established. A solute with finite solubility can yield a saturated solution when it is added to a solvent in an amount exceeding its solubility, resulting in a heterogeneous mixture of the saturated solution and the excess, undissolved solute. The equation that describes the equilibrium between solid calcium carbonate and its solvated ions is. In this episode of Crash Course Chemistry, we learn about precipitation, precipitates, anions, cations, and how to. An example of a precipitation reaction is N a2SO4(aq) + BaCl2(aq) 2N aCl(aq) + BaSO4(s) N a 2 S O 4 ( a q) + B a C l 2 ( a q) 2 N a C l ( a q) + B a S O 4 ( s). But when two ions find each other that form an insoluble compound, they suddenly fall out of solution in what's called a precipitation reaction. Recall from the chapter on solutions that the solubility of a substance can vary from essentially zero ( insoluble or sparingly soluble) to infinity ( miscible). A lot of ionic compounds dissolve in water, dissociating into individual ions. This section applies previously introduced equilibrium concepts and tools to systems involving dissolution and precipitation. The chemical reaction between potassium chloride (KCl) and silver nitrate (AgNO3), and solid silver chloride (AgCl) is the precipitate or the insoluble salt. 1 In the following section, we will examine the reaction that occurs when a solid piece of elemental magnesium in placed in an aqueous solution of copper (II) chloride: (8.5. An understanding of the factors affecting compound solubility is, therefore, essential to the effective management of these processes. It might be helpful to look at a few more examples. These equilibria underlie many natural and technological processes, ranging from tooth decay to water purification. Solubility equilibria are established when the dissolution and precipitation of a solute species occur at equal rates. Carry out equilibrium computations involving solubility, equilibrium expressions, and solute concentrations To balance the equation, we need a two in front of NANO3.In other words, break all of the strong electrolytes into the ions they form in aqueous solution. Write chemical equations and equilibrium expressions representing solubility equilibria Write the equation in terms of all of the ions in the solution.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
